An Alternative Strategy for Fighting SARS-CoV-2 Infection
The severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2)-caused coronavirus illness 2019 (COVID-19) has been more than an infectious disease outbreak. It not only put an unprecedented burden on healthcare institutions, the global economy, and society, but it also pushed the global research community to explore novel treatment possibilities like never before.
While multiple drugs that had already been tested in other conditions with similar infection patterns, that is:
- Ebola virus,
- MERS-CoV was deployed during the first hit of the SARS-CoV-2 outbreak.
At this pandemic stage, specific methods based on physiopathology to prevent and treat COVID-19 are expected to emerge. COVID-19’s therapeutic targets and treatments have been divided into “target virus” and “target host.” Based on viral and host factors that occur at the levels of DNA, RNA, and proteins, involving both classic and novel critical signalling pathways, and even including promising epigenetic mechanisms that contribute to SARS-CoV-2 infection, a comprehensive analysis of therapeutic targets has been conducted.
Acute respiratory distress syndrome (ARDS) can result from cytokine storm syndrome (CSS), which accounts for the majority of COVID-19 deaths. Interleukin 6 (IL-6) has been identified as a critical hallmark of the cytokine storm, and clinical usage of the IL-6 inhibitor tocilizumab has shown promise in the treatment of COVID-19 patients.
A Current Focused Area on Fighting the COVID-19
1. Cytokines
Acute respiratory distress syndrome (ARDS) and cytokine storm syndrome (CSS) may emerge as COVID-19 develops. CSS is linked to the progression of various severe diseases and entails a rapid increase in proinflammatory cytokine levels in response to a stimulus. In COVID-19 patients, CSS is marked by significant increases in the plasma levels of proinflammatory cytokines, including:
- IL-6,
- IL-1β,
- IL-2,
- IL-8,
- IL-17,
- Granulocyte colony-stimulating factor (G-CSF),
- Granulocyte-macrophage colony-stimulating factor (GM-CSF), IP-10,
- Monocyte chemoattractant protein (MCP)-1,
- Macrophage inflammatory protein (MIP)-1α,
- Tumour necrosis factor (TNF)α leads to the recruitment of immune cells through a positive feedback loop, eventually forming a cytokine storm.
Clarifying the mechanism of CSS in COVID-19 and identifying the significant cytokines involved will aid in the development of more efficient CSS-blocking treatments and would be crucial in the treatment of severe cases.
2. Microbiome as a Target for SARS-CoV2
Because the gut microbiota is changed in COVID-19 patients, probiotic administration may help restore the gut microbiota and, as a result, maintain a healthy gut-lung axis while reducing pathogenic bacteria translocation over the gut barrier and the risk of secondary bacterial infections. Because cytokines storm develops in patients with severe COVID-19, immune-modulatory effects of probiotics may help prevent life-threatening COVID-19 consequences such as acute lung damage, acute respiratory distress syndrome, and multiple organ failure.
3. Dietary habits
Several studies have shown that eating habits and the amount of food consumed can influence the microbiome of humans. Developing-country diets are typically higher in fibre than current Western diets, which are commonly processed and maintained in cold storage. Varied groups with diverse diets may have different microbiome patterns, which are now well understood.
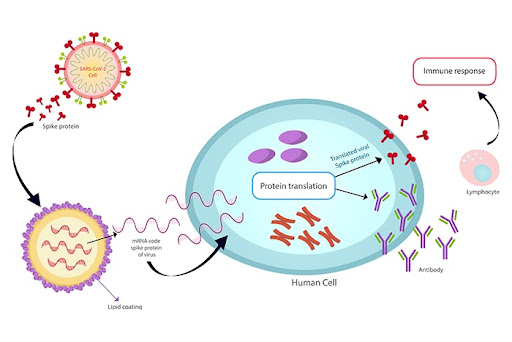
4. Vaccines of SARS-CoV-2
Since the rapid, unprecedented admission of the first SARS-CoV-2 vaccine candidate on March 16, 2020, vaccines underwent preclinical evolution, and 100 others are undergoing clinical trials. EUA has licenced or approved vaccines for SARS-CoV-2 around the world. These vaccines are of four types: viral vector-based vaccines, RNA-based vaccines, inactivated virus vaccines, and protein subunit vaccines.
Due to the temporary halt in clinical studies, virus-like particle vaccinations may require more time to assess their efficacy and safety. Sputnik V and three inactivated vaccines are licenced in Russia, and Ad5-nCoV and three inactivated vaccines are licenced in China. BNT162b2, mRNA-1273, and Ad26 are among the EUA vaccines. In America, COV2.S is used; in England, Covishield is used; in India, Covaxin is used; and in China, ZF2001 is used.
5. S protein in SARS-CoV variants
Increased transmissibility was linked to Sipke protein mutations, which played a crucial role in the early stages of the COVID-19 pandemic. However, this mutation had a minor effect in neutralising the sera of vaccinated people and COVID-19 patients.
SARS-CoV-2 variants with greater transmissibility have recently arisen worldwide, posing a threat to viral control and increasing concerns that unforeseen and constant alterations may weaken the pandemic’s current efforts. As a result, the three principal SARS-CoV-2 variants responsible for the outbreak, as well as other potential treatments to prevent reinfection by new variants, have been summarised.
- When SARS-CoV-2 variant B.1.1.7 (UK delta variante), also known as 501Y.V1 or variant of concern 202012/01, initially appeared in England, it increased COVID-19 cases. Except for D614G, it possesses eight S protein mutations.
- SARS-CoV-2 P.1 (501Y.V3) and B.1.351 (501Y.V2), also known as the South Africa variation and the Brazil variant, have the most robust infectious capacity.
Previous research showed no significant variations in virus entry kinetics, virus-cell and cell growth fusion efficiency, or Spike protein stability between SARS-CoV-2 WT and variants B.1.1.7, B.1.351, and P.1.
6. Monoclonal antibodies
mAbs have been shown to be resistant to B.1.1.7, B.1.351, and P.1 variations in various studies. The B.1.1.7 variation, which is primarily given by 144 deletions, is resistant to neutralisation by NTD supersite-directed mAbs.
The R246I and 242–244 deletions play a significant role in B.1.351 resistance. The NTD supersite includes all 144 and 242–244 omissions and R246I.
P.1 has NTD mutations (R190S, D138Y, P26S, T20N, and L18F) rather than NTD deletions, which could affect mAb binding. These EUA mAbs that target RBD are particularly important in B.1.351 and P.1 resistance.
Recently, existing mAbs, such as casirivimab and imdevimab, have only shown limited protection against SARS-CoV-2 Virus subtypes. However, the efficacy of mAbs against variations has to be investigated further due to the lack of large-scale clinical evidence. Meanwhile, global virus genomic surveillance and promotion of next-generation antibody treatments, particularly the combination that targets various antigen epitopes, should emphasise their importance.
7. Anti-inflammatory cytokines
In patients with COVID-19, other proinflammatory cytokines or receptor inhibitors, such as IL-1 and IL-6 receptor inhibitors, demonstrated significant survival improvements. Anakinra is a recombinant human IL-1 receptor antagonist that inhibits IL-1 production. It’s been approved to treat rheumatoid arthritis and cryopyrin-related rare disorders. In several case reports, patients with cytokine release syndrome or macrophage activation syndrome, which were assumed to be one of the reasons for ARDS in COVID-19 patients, had positive outcomes.
Conclusion
There is still a lack of knowledge about the long-term effects of COVID-19 illness. The current outbreak has piqued the curiosity of researchers searching for viable antiviral medications and prevention techniques. There is still an urgent need to characterise bacterial co-infections in this rapidly growing field. Co-infection with different bacteria has had a significant impact on human health.
Scientists concentrate on harmful viral proteins based on previous experiences with SARS-CoV and MERS-CoV. As a result, repurposing medications may be the best option, as it can reduce drug development time by demonstrating efficacy and safety in clinical trials.
Get in touch with Helvetica Health Care or visit our website to explore our CELLKINES™ product line of Human Interleukin-2 (IL-2), also referred to as T-CELL GROWTH FACTOR (TCGF).
ABOUT THE AUTHOR
“Michael Moore is a chartered marketeer and member of the Institute of Marketing with over 35 years experience in the Biotechnology industry, with particular expertise in International Business Development, distributor management, and international tender business.“
