The way to Antibody Validation standards and its Challenges
An Antibody is one of the most universally used reagents in science research, clinical tests and diagnostics. Regardless of their well-known use and the considerable investment of time and money that their benefits represent, no standard guidelines define how antibodies should be validated before use.
What is Antibody validation?
Validation is an experimental proof and documentation that a definitive antibody is suitable for a predetermined application or purpose. In simpler terms antibody validation can be defined by the following points:
● Proving specificity which means the ability of an antibody to differentiate between different antigens.
● Proving the application specificity in which it is going to be used.
● Proving similarity which means the potency with which an antibody binds an epitope.
● Proving reproducibility.
However, while the above definition of antibody validation is practical, the common application or implementation of a standardized process remains a significant concern.
Antibody Validation Standards
The success of any immunodetection experiment relies on the quality of the antibodies which are employed. Few antibodies may be working in specific applications, and poorly in others. Thus, the user must know which applications an antibody is validated for and those for which it failed validation.
1. Antibody Information: Detail regarding the immunogen’s character to generate an antibody can provide an honest indication of whether the antibody will work effectively within the specific application.
2. Target Information: By studying the background information surrounding the target protein, researchers can gain a transparent understanding of the likelihood of detecting it within the chosen sample material. Target information provides a wealth of data regarding protein expression at the tissue level and within cells. These powerful resources are often used freely to explore expected patterns of tissue or cellular localisation.
3. Species Validation: In many cases, an antibody raised against a person’s antigen will also recognise that very same antigen in samples derived from other species. The details of the species against which the antibody has been tested and located to figure will be listed on the merchandise datasheet. Often, species against which the antibody has been tested and discovered to be unsuccessful will also be detailed, alongside predicted untested species.
4. Application Validation: Product datasheets will typically list all the applications during which an antibody has been tested, with the foremost standard samples of these being immunocytochemistry, western blot, and immunohistochemistry. The datasheet should specify the sample type against which the antibody has been validated for IHC.
5. Storage and Handling Information: Most antibodies are provided as solutions, which might include tissue culture supernatant, ascites fluid, or a purified antibody product during a defined buffer, others are supplied during a lyophilised format requiring reconstitution. Liquid antibodies should be centrifuged briefly before being opened, to stop loss or contamination of the merchandise.
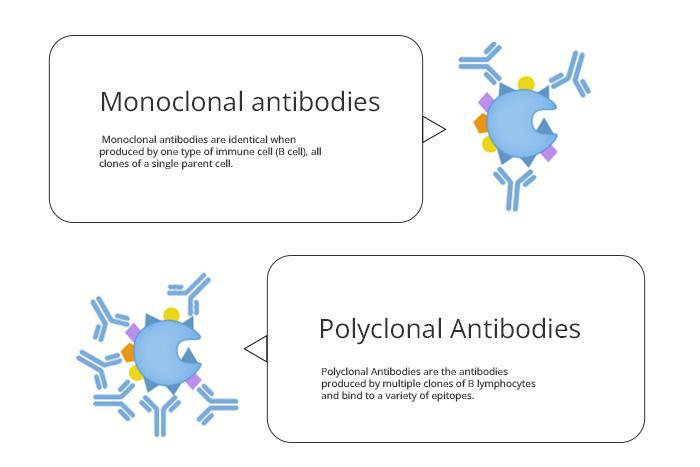
6. Lot Validation: Each unique lot of an antibody should be furnished with lot-specific data, including the concentration of that lot and validation data for each of the applications during which it’s been tested. It should never be assumed, especially within polyclonal antibodies, that each antibody batch will show an identical pattern of behaviour.
7. Quality Control: The development and manufacturing processes are subject to rigorous internal control and quality assurance measures. All of our antibody products are furnished with a comprehensive Certificate of analysis and product Information Sheet.
Challenges of Antibody Validation:
● Unfortunately, antibodies are often capricious reagents and need individual care. Therefore, not all stabilisation and storage protocols apply to all or any antibodies. Validation of stability under specified conditions is beneficial if an antibody should be used for an extended time.
● Low-affinity antibodies often cause weak signals, nonspecific background, and high costs because of the high concentrations needed for the assays.
● Reagents without proper documentation might cause a waste of some time and money. Sometimes it’s better to develop your own, well-documented antibody, on twiddle a product, from which nearly nothing is known and eventually are getting to be found as unsuitable.
● Low concentrations cause weak signals and high concentrations to nonspecific interactions. Any wrong information about antibody concentration might make the event or optimise much more difficult and laborious.
● The more Cross-Reactivity data available for an antibody or immunoassay, the higher the performer evaluates the usability during a selected application context.
● If the target isn’t defined correctly, it cannot be decided, whether the binder is selective enough or not. A low specification of the target analyte is the foremost frequent reason for the complete failure of an immunoassay.
● The high cost of the various antibody reagents, poor stability can cause an enormous financial burden and frustration. Information about the storage conditions of a specific antibody could also be an honest indication of professional development and control.
Contact Helvetica Health Care for more information on Antibody Validation Standards and Challenges.